Thanks to its sensor technology, the ABL800 FLEX measures creatinine with dependable lab quality with whole blood. In addition, the analyzer reports eGFR for effective chronic kidney disease screening and prevention of contrast-induced nephropathy.
About creatinine
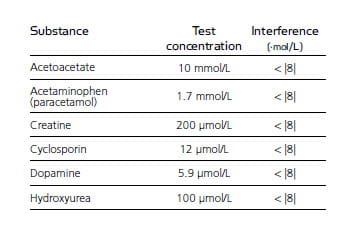
The concentration of creatinine provides information on kidney function and muscle mass. There is a poor track record for creatinine in other measurement systems1, and a fluctuation in results due to interference is one of the main problems for creatinine measurements.
Suppression of 62 interfering substances ensures reliable results
Through its choice of sensor technology, Radiometer has been able to suppress interference from a significant number of substances.
In total, 62 different endogenous and exogenous substances were identified and subjected to an interference test protocol on creatinine. None of them showed detectable signs of interference 2.
eGFR on the ABL800 FLEX meets NKDEP requirements
Estimated Glomerular Filtration Rate (eGFR) is recognized to have great importance in the early detection and management of chronic kidney disease. The eGFR indicates how effectively the kidneys clear out the substrates from the body. Besides creatinine, the eGFR algorithm is based on age, gender and race.
Because mild and moderate kidney injury is poorly inferred from serum creatinine alone, The National Kidney Disease Education Program (NKDEP) “strongly encourages clinical laboratories to automatically report eGFR when serum creatinine is reported” 3. The purpose of NKDEP is to reduce the morbidity and mortality caused by kidney disease and its complications.
The ABL800 FLEX eGFR formula meets the NKDEP requirements and the analyzer reports eGFR following the Modification of Diet in Renal Disease (MDRD) Study equation.
Standardization of creatinine measurement
ABL800 FLEX creatinine measurement follows the standardization recommended by NKDEP using the recommended reference method IDMS (sotope dilution mass spectrometry).
NKDEP has defined boundaries for combinations of systematic bias and imprecision in serum creatinine measurements4.
For more on what to consider before instituting whole-blood creatinine/eGFR for screening of chronic kidney disease and prevention of contrast-induced nephropathy, listen to the on-demand webinar The New Role of STAT Creatinine Testing presented by Dr. Brad Karon of the Mayo Clinic.
1. Myers GL, Miller WG, Coresh J et al. Recommendations for improving serum creatinine measurement: a report from the National Kidney Disease Education Program. Clin Chem 2006; 52: 5-18.
2. Andersson, AC et al. "Interference testing of the creatinine sensor in the ABL837 FLEX analyzer." Point-of-care 6.2 (2007): 135-43.
3. National Kidney Disease Education Program and NKDEP. Suggestions for laboratories. revised Jan 2007.
4. Skurup, A, T Kristensen, and G Wennecke. "New creatinine sensor for point-of-care testing of creatinine meets the National Kidney Disease Education Program guidelines." Clin Chem Lab Med 46.1 (2007).

